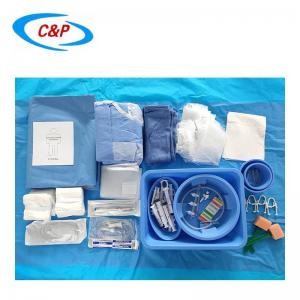
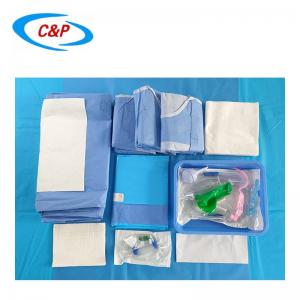
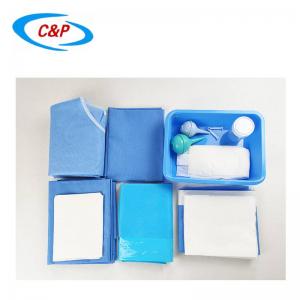
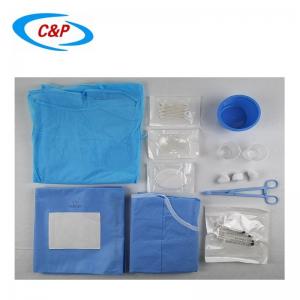
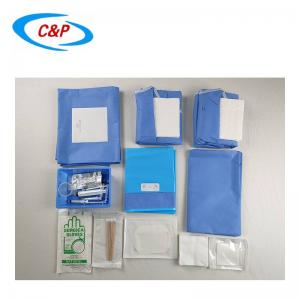
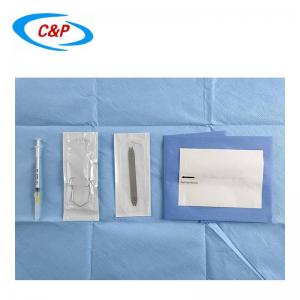
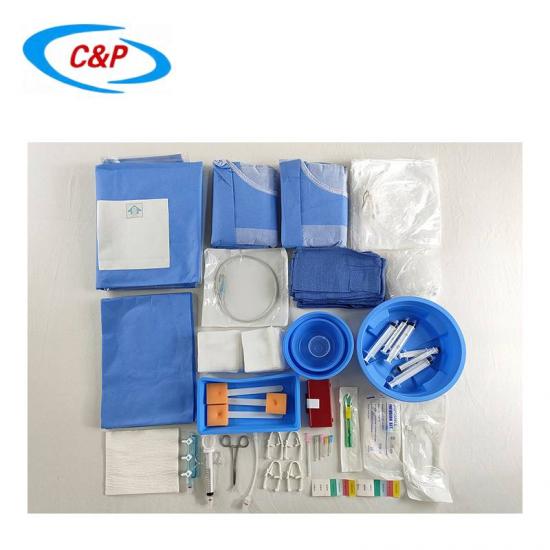
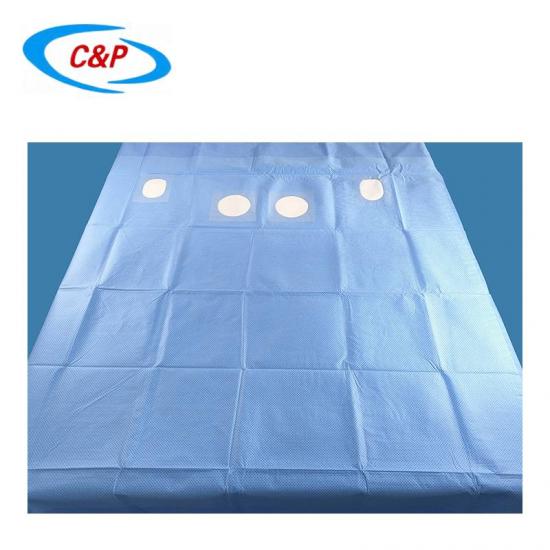
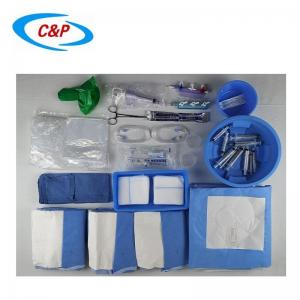
Our sterile angiography surgical pack is an advanced, disposable surgical pack system specifically designed for angiography procedures, including cardiac angiography, cerebral angiography, peripheral angiography, and endovascular interventional procedures. Made from advanced medical-grade materials, the pack establishes a reliable sterile barrier, effectively isolates the surgical site, manages fluids, and optimizes the surgical field of vision—essential for precise, safe, and efficient angiography surgery.
PRODUCT PARAMETERS
|
Name |
Angiography Drape Pack |
|
Material |
SMS, SMMS, SMMMS, PP, PE, Spunlace Nonwoven |
|
Thickness |
20 -100gsm |
|
Color |
Blue, green or as request |
|
Details |
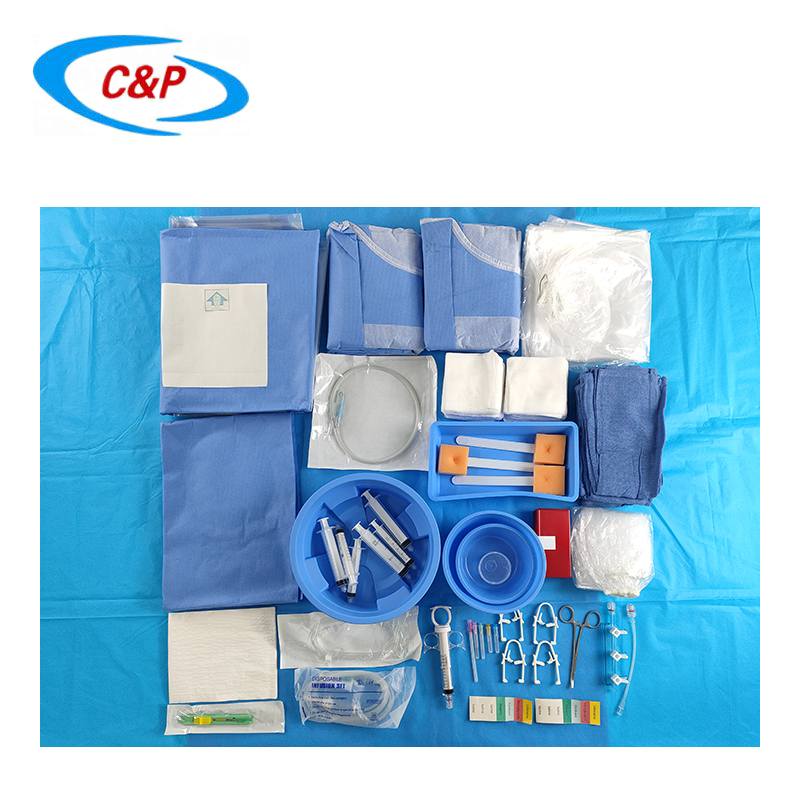
2pc Band Bag Dome 91 cm x 122cm
1pc Band Bag Dome 45 in (114 cm)
1pc Mosquito forceps curved 5"
1pc Needle 18Gx 1 1/2 Reg Bev
1pc Needle 21Gx 1 1/2 Reg Bev
1pc Needle 23Gx 1 1/2 Reg Bev
1pc Needle 25Gx 1 1/2 Reg Bev
1pc Bowl, 500 cc
1pc Bowl, 1000 cc
1pc Tray, 9"x5"x 2"
50pc Gauze swabs 4"x4", 12Ply
1pc Manifold, 3 port OFF
1pc Sharps holder
1pc Pressure Monitoring line 6" M/F
1pc Pressure Monitoring line 180cm
1pc IV line, 180cm large bore
1pc IV line, 180cm non-vented
1pc Control syringe 12ml LL
1pc Syringe label sheet
1pc Chlorhexidine prep 10.8 ml, Clear Non-Sterile
1pc Entry Needle 18Gx7cm
1pc Guide wire J Tip PTFE Coated 0.035 x 150cm
1pc Reinforced Angiography drape Fem Rad, 240x380cm, w/4 Fen
2pc Gown, XL
2pc Hand Towel 30x40cm
1pc Set up Cover, 135cmx200cm
1pc Back Table Cover, 150cm x 200cm
6pc Blue Towel
1pc Bowl Guide wire 2500CC
1pc Control Syringe 12ml
3pc Syringe 20 cc LL
3pc Syringe 10 mL LL
3pc Sponge Stick 8"
1pc Safety Scalpel #11 Blade
1pc Cup, 60cc
2pc Label Manifold
|
|
Packing |
1pc/sterile pack, 5pcs/ctn, ctn size:60x40x50cm or as request |
|
Shipping Port |
Shanghai, Ningbo |
|
OEM |
Material, Size, LOGO, Package or other specifications |
|
Quality Certification |
CE, ISO13485, EN13795 |
|
Sterile |
EO Sterile |
PRODUCT DISPLAY
FEATURES
I. Safe Materials, Suitable for Medical-Grade Surgery Needs
Using medical-grade disposable non-woven fabric as the core material, it is non-allergenic and non-irritating, allowing direct contact with surgical wounds and mucous membranes, effectively blocking external bacteria, dust, and other contaminants.
II. Sterility Guarantee, Reducing the Risk of Surgical Infection
The entire production process employs sterile technology. From raw material procurement and processing to packaging and sealing, everything is completed in a Class 10,000 cleanroom, strictly adhering to medical sterility standards.
III. Precise Configuration, Fitting Angiography Surgery Scenarios
Customized components are designed for the core procedures of angiography surgery, eliminating the need for additional assembly and improving surgical efficiency.
IV. Single-Use, Eliminating Cross-Infection
Using a single-use design, it can be directly and uniformly recycled after surgery, eliminating the need for washing, disinfection, and reuse, fundamentally eliminating the risk of cross-infection and meeting the core requirements of modern medical infection control.
V. Convenient Operation, Improving Surgical Efficiency
The packaging design is user-friendly, with internal components categorized and clearly labeled for easy access by medical staff.
VI. Compliance and Quality Assurance
Our products strictly adhere to the ISO 13485 medical device quality management system standard and relevant national medical device regulations. They have obtained international authoritative certifications such as CE, and their quality meets the access requirements of the global medical market.
COMPANY PROFILE

Hefei C&P Nonwoven Products Co.,Ltd. is a private company established in 2007, C&P located in Hefei city, capital of Anhui, C&P is a manufacturer and supplier of surgical drapes&packs, surgical gown, isolation gown, masks, equipment covers and other disposable medical products. We assemble packs specially for all kinds of surgeries. Our 3000 square meter 100,000 grade standard clean room (ISO8) space and more than 100 employees aiming to flexibility and cost effective efficiency.
OUR ADVANTAGES
1. Rich production experience: With 18 years of experience in the production of medical consumables, valuable knowledge and expertise have been accumulated in this field. Enable us to better understand the needs and requirements of the industry, in order to produce high-quality products.
2. Compliance with international standards: CE, ISO13485 certificate is an internationally recognized standard for quality management in the medical device industry. This certification demonstrates that our products and processes comply with strict safety and efficacy regulatory requirements, ensuring that our product is suitable for its intended use.
3. ISO 8 clean room: ISO 8 clean rooms provide a highly controlled environment with limited particulate matter in the air. This cleanliness is crucial for the production of certain medical consumables, as it can reduce the risk of product contamination and ensure sterility.
4. Good performance: Exported to more than 20 countries.
5. Design team: We have a professional design team can do OEM&ODM.
OUR CERTIFICATE

OUR EXHIBITION

RECOMMENDED PRODUCTS